Canadian Customers: Take a look at our simplified Returns Process

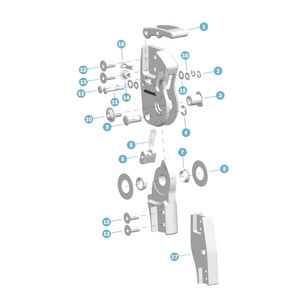




ARTICUL. UNILATERALE GENOUX, TITANE
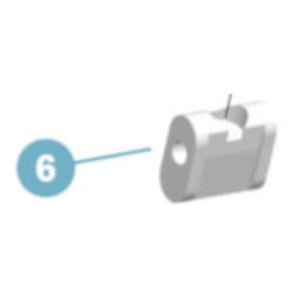
Item #: 17LK3=L16-TTitanium system knee joint with wedge lock
Détails du produit
The lightweight construction of the 17LK3 Unilateral Knee Joint makes it particularly attractive. This system knee joint with a wedge lock comes with a temporary switch for releasing the joint for activities such as training on a therapeutic bike.
Indications
Partial or total paralysis of the leg muscles. Indication must be determined by a physician.
About the Unilateral Joint System
With the Ottobock Unilateral Joint System, you can experience all the benefits of a unilateral system combined with the confidence of selecting the appropriate weight classification. The unique unilateral design means that you can stock less inventory while still accommodating a wide range of patients. Plus, you avoid the worry of a medial joint and have the option to accommodate a weight or functional need by using the joints in tandem. You’ll find that the system’s modular knee and ankle joints make it easy to update as your patient’s needs change. The components come in a range of sizes designed to target your patient’s needs – accommodating up to 220 lbs / 99 kgs in a single upright configuration and 350 lbs / 159 kgs in a double upright KAFO. In addition, the joints work with a MTP to floor measurement of up to 55 cms.
- Modular, multifunctional joint system
- Lightweight construction
- Available in four system sizes for both adult and pediatric fittings
- Can be used for prepreg and lamination resin techniques
- Weight classification for unilateral and bilateral use simplifies joint size selection
- Can be used on medial side of knee, lateral side, or both
- Temporary switch releases the joint for training on a therapeutic bike
Accessories
Pièces de rechange
Documents
Guide de sélection des orthèses cruro-pédieuses.
Information produit
Document PDF | Date de publication : 24 avr. 2026